
Properties of water
- Two parts of water are hydrogen and oxygen.
- In water there are two hydrogen atoms and one oxygen.
- Atoms are the smallest thing ever to existed.
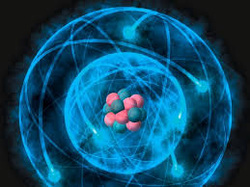
- Protons and Neutrons
- Protons have a positive charged.
- Electrons have a negative charged.
- Neutrons have a neutrally charged.
- Atoms are usually neutrally charged.
- Each H (hydrogen) atoms and one O (oxygen) atoms combine to form a water
molecule. - Each H shares one of its single molecule with the O.
- The O shares one of its electrons with the H.

Water is polar molecule
Hydrogen Bonding- the weak
attraction between the weakly positive hydrogen atoms and the weakly
negative atoms of a different molecule.
Water is a universal solvent - The polarity of the water gives an
ability to dissolve most compounds.
Positive hydrogen side of H2O is attracted to the negative parts of the
compound.Negative is ALWAYS attracted to
positive.
Water is attracted to other water molecules which
is also called Cohesion.
Adhesion- Is
when water is attracted to other
materials.
Surface Tension-
Is when Water molecules
create a surface for light objects and
animals.
States of matter
Hydrogen Bonding- the weak
attraction between the weakly positive hydrogen atoms and the weakly
negative atoms of a different molecule.
Water is a universal solvent - The polarity of the water gives an
ability to dissolve most compounds.
Positive hydrogen side of H2O is attracted to the negative parts of the
compound.Negative is ALWAYS attracted to
positive.
Water is attracted to other water molecules which
is also called Cohesion.
Adhesion- Is
when water is attracted to other
materials.
Surface Tension-
Is when Water molecules
create a surface for light objects and
animals.
States of matter
- The gas state of water is water vapor.
- The liquid state of water is water.
- The solid state of water is
ice.
